Tamoxifen
Tamoxifen is the most widely prescribed cancer drug worldwide for women with early-stage estrogen receptor positive (ER+), HER2-negative breast cancer. After finishing their treatment for the cancer, they are prescribed tamoxifen to prevent relapse. A 5- to 10-year treatment delays local and distant recurrence, prolongs overall survival, and reduces the incidence of contralateral breast cancer.
The standard dose of tamoxifen, 20 mg/day, is given to all women independent their BMI, inter-individual variation, liver disease and other drugs. This could either lead to overdose, causing side effects or too low dose. According to several studies, Tamoxifen adherence to therapy may be as low as 50%, because many women suffer from severe side effects and discontinue medication. Poor adherence and early discontinuation are both associated with an increased risk of breast cancer recurrence.
Therapeutic Drug Monitoring
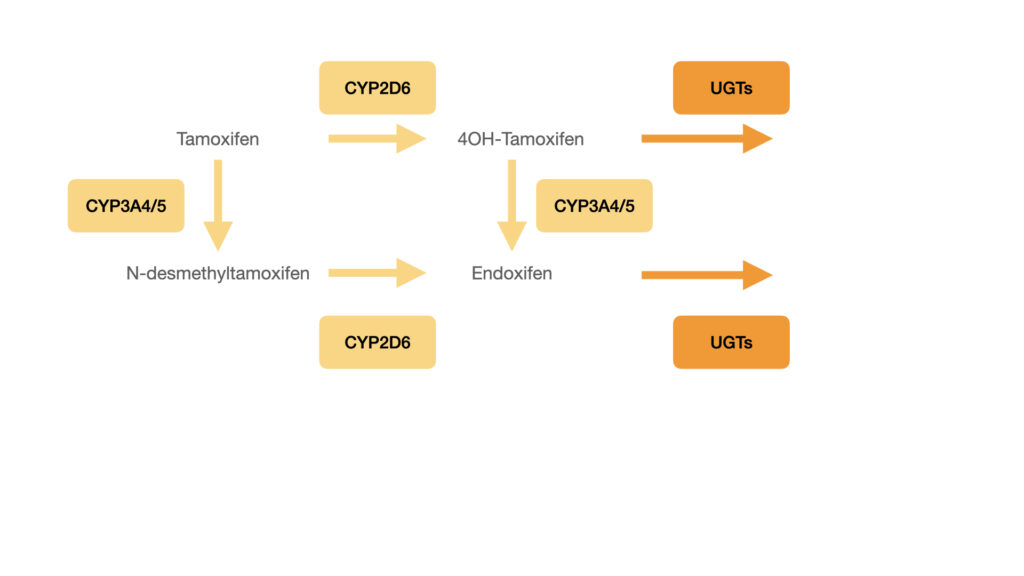
An individualized prescription based on the actual concentrations of the drug and its primary metabolites in the blood would increase the possibility of receiving optimal treatment, reduce the side effects and enhance quality of life. However, the conventional method of tamoxifen therapeutic drug monitoring is complicated and expensive, and therefore not widely used.
redhot’s rhelise™ kit and the tamoxifen analysis kit are ideally suited for therapeutic drug monitoring and developed to provide women under tamoxifen treatment with an easy to use at-home test and the analysis lab with a straightforward and robust method for LC-MS/MS quantitative determination of tamoxifen, Z-endoxifen and 4–hydroxy tamoxifen in whole blood.
Article number | Description |
|---|---|
30-2001 | Tamoxifen kit including column, 100 determinations |
30-2002 | Tamoxifen kit, 100 determinations |
